The Physics ABC

The Physics Alphabet
Most people leave High School with the view that physics is hard. It is beguiling, befuddling and bemusing; many people are left with the feeling that physics is the mutant hybrid of a foreign language and complex mathematics. Physicists themselves, with one or two notable exceptions, are notoriously bad at communication - often forgetting that their audience are not familiar with the lexicon of quantum mechanics.
The thing is, once you get your head around the complicated language used in physics, you find out that it isn't as difficult as you think. Whilst physics uses maths and equations to describe what is going on in the universe, simple analogies can take us a long way.
This guide aims to clear up a few of the key terms you will come across in the field of Physics. The alphabet was written by my Yr 9 pupils (13/14yrs old) as a revision activity - I've added a bit more detail as well as some helpful videos and diagrams. Enjoy!
How Small is an Atom? (TED Video)
A is for Atom
The atom is the basic unit of an element. For many years it was thought that atoms were the basic unit of the universe. It was then discovered that atoms were made up of even smaller sub-atomic particles:
- Protons - one of two particles in the nucleus of the atom, these increase by one as you move across the periodic table. Protons are positively charged and determine the properties of the element.
- Neutrons - these are negatively charged particles in the nucleus of the atom. The numbers of neutrons differ depending on the isotope of the element. Different isotopes therefore weigh different amounts. Hydrogen does not have any neutrons in it's nucleus.
- Electron - these tiny sub-atomic particles have a charge equal and opposite to 1 proton. They whiz around the outside of the nucleus in a cloud.
Over 99% of the mass of an atom is contained in the nucleus, with a typical atom measuring just a few tenths of a nanometer across. Most of an atom is actually empty space - a mind-boggling thought when you think that all matter, from your chair, table and pc screen is made up of tiny particles that are mostly empty!
The name for atom comes from the Greek for 'indivisible' - the name has sort of stuck, despite finding out that an atom can be further divided!
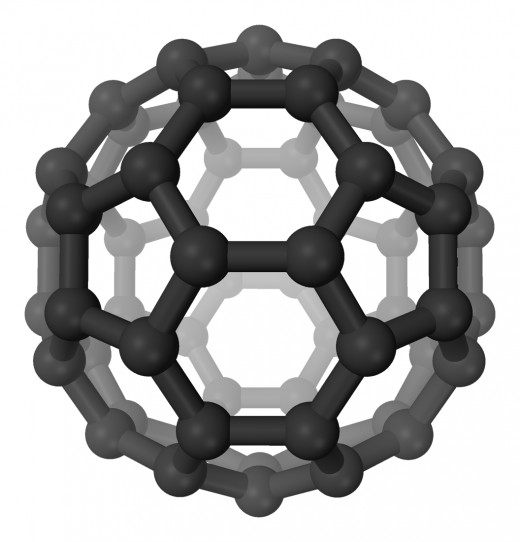
B is for Buckminster Fullerene
Eh? What? There they go again, those pesky physicists making up words. Buckminster Fullerene (C60) was discovered in 1985. Each molecule has 60 carbon atoms that arrange themselves to form a hollow sphere. These 'buckyballs' are said to be allotropes (like graphite, diamond, graphene) - molecules made from the same atom, in the same state at room temperature, but with their atoms arranged differently.
These 'fullerenes' are being used as cages for delivery of drugs - this means they can be used to target cancer cells much more accurately and effectively. The balls can also be joined together to form carbon nanotubes. These tubes are some of the strongest structures on Earth - they are woven into fibres to make carbon fibre - used to create materials that are very strong but also very light (think F1 racing cars)
C is for Coulomb
Coulombs (C - thats capital C btw, small c is the speed of light in a vacuum - 186,282 miles per second if you are interested.) are the units of charge. Charge is simply an amount of electricity. The number of coulombs tell us how much electricity is transferred in one second, by a current of 1amp.